Are you suffering from dry eye disease?
Are you suffering from dry eye disease?
Dry eye symptoms include:
- Redness
- Burning
- Itching
- Fluctuation in vision
- The feeling of sand or grit
- Contact lens discomfort
- Light sensitivity
- Watery eyes
- Tired eyes
If you have two or more symptoms, you may have Dry Eye Disease and should discuss your symptoms with your doctor.
What is dry eye disease?
Dry eye disease affects the cornea or surface of the eye. Tears spread across the cornea with every blink. The amount and quality of one’s tear are essential to keep your eyes comfortable and clear. In addition, there are muscles, nerves, glands, tear ducts, and connective tissue that must work together to keep the eyes moist. Dry eye disease usually occurs when one of these factors is out of balance, causing imbalance to other factors, perpetuating the cycle of dry eye disease. Dry eye disease is a chronic condition with no one solution.
According to research, over 16 million Americans have dry eyes. The risk of developing dry eye disease increases with age. Women are more likely to develop the condition compared with men. However, dry eye disease is becoming increasingly common in younger people, with more device use like smartphones, laptops and tablets, than ever before.
Dry eyes can even impact your quality of life.
Dry eye disease is often overlooked but can have serious short-term and long-term effects on those who develop it. Short-term effects can be uncomfortable and annoying and include constant eye irritation, blurry vision, regularly tired eyes and difficulty wearing contact lenses. Simple daily activities like reading, watching television, using the computer and mobile phones can be difficult when struggling with dry eye. Long term, dry eye can lead to permanent vision impairment if not taken care of and managed well. Thus, diagnosing and treating dry eye disease during its early stages is essential.
Why are tears important for the eyes?
Tears play an essential role in protecting and keeping your eyes healthy. They wash away unwanted debris and keep the surface of the eye clear. Natural proteins from tears help fight germs and things that cause an eye infection.
Tears have three layers — the outer lipid layer, the middle aqueous layer, and the inner mucin layer. Each layer serves a purpose.
- The inner layer is made up of mucus that coats the surface of the eye. The mucin layer binds tears to the eye and is also responsible for spreading the water layer evenly over the surface of the eye.
- The middle layer, which is also the thickest layer, is primarily composed of an aqueous or watery layer. This layer, which is more like a saline solution, contains vitamins and minerals that keep the eye nourished.
- The outermost layer of the tear is an oil or lipid-based layer. It seals the tear film, keeping the surface of the tear smooth and preventing the other layers from evaporating. In other words, the oily layer keeps your tears from drying up too quickly.

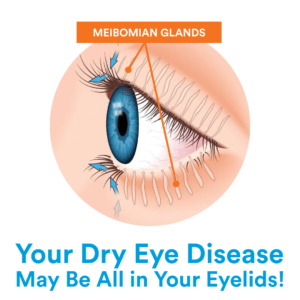
The aqueous glands within the eyelids produce the watery layer of our tears, and the Meibomian glands along the upper and lower eyelid’s margins secrete the lipid or oily layer.
Dry eyes can occur when there is a loss of natural balance (either from external or internal problems) with any of these layers or when the glands get clogged, becoming dysfunctional and permanently damaged over time, leading to severe dry eye symptoms.
What are the causes of dry eyes?
The two leading causes of dry eyes are rapid evaporation of tears caused by a deficiency in the oil layer and reduced quantity of tears produced by the glands. Often, there is overlap between these two leading causes of dry eyes. Several factors can affect tear production or the condition of the eye in general, including but not limited to:
Incomplete blinking
Reading, staring at a smartphone or computer, driving, and other similar activities require concentration, leading people to blink less often, which in turn causes increased tear evaporation.
Age
The amount and quality of tears the eyes produce decrease with age. Dry eyes increase in prevalence for people aged 55 and above.
Gender
Due to hormonal changes, pregnancy, contraceptives and menopause, women have a higher chance of developing dry eyes.
Cosmetics
Some cosmetic ingredients can be irritating to the eyes and cause dry eyes. Hypoallergenic, vegan, organic or sensitive mascara may help if this is the cause or if cosmetics exacerbate the cause.
Eyelid problems
Conditions such as ectropion (out-turning of the lids), entropion (in-turning of the lids) can produce more exposure or friction and cause the normal tear film to dry faster.
Recent procedures
Refractive eye surgery, like LASIK, can cause decreased tear production due to a permanent change to the front surface of the eye. For some people, dry eye symptoms related to eye procedures can be temporary. Others can have persistent symptoms.
Medications
Most medicines have side effects, and some of these can affect tear production. Medications to manage blood pressure and Parkinson’s disease can reduce tear production. Other medications including, antidepressants, sleeping pills, heartburn tablets, drugs for acne, birth control, hormone replacement and cold and allergy pills, can reduce tear production.
Health conditions
Autoimmune diseases like rheumatoid arthritis, Sjogren’s syndrome, scleroderma, lupus, and thyroid disease can directly affect the glands responsible for tear production. These conditions can cause imbalances in the three main layers of the tear — mucus, oil and water. Vitamin A deficiency can also decrease tear production.
Indoor and outdoor environmental conditions
Your eyes (and tears) are the most exposed parts of your body. Exposure to a dry environment, whether indoor or outdoor, can affect the eyes. People who live in places with dry climates are more prone to having dry eyes. Exposure to wind, smoke and fumes can also aggravate your eyes. Dry indoor environments include air-conditioned rooms, hospitals, airplanes and workplaces.
Lifestyle
Contact lenses can also irritate the surface of the eye and contribute to dry eyes. Other activities like cigarette smoking, alcohol consumption, and not getting enough sleep can also affect the dryness of your eyes.
Dry Eye Testing
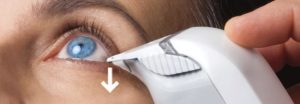
In the past, doctors had to rely on your description of the symptoms and dye testing to diagnose this disease and had no good way to measure whether it was getting better. Now with TearLab technology, you can be tested and given objective results immediately. Your doctor will take a sample of tears from each eye and use the TearLab to measure osmolarity, which evaluates the aqueous or watery layer of the tear film. The sample required is no larger than the period at the end of this sentence and only takes seconds to collect. We all have salt in our tears. If the watery layer evaporates, it leaves us with too much salt and high osmolarity. In moments we know the osmolarity level in your tears. (> 300 is somewhat dry. > 305 usually causes symptoms).
We also use InflammaDry®, an in-office, rapid result test that detects elevated levels of matrix metalloproteinase 9 (MMP-9), an inflammatory marker consistently elevated in the tears of patients with dry eye disease. This test evaluates the possible inflammatory component of dry eye. If inflammation is present, your doctor may prescribe specific drops for treatment.
This advanced testing helps identify the cause(s) to better address the root of the problem.
Treatment
Dry eyes are often managed, not cured. However, research and technology have given us significant treatment improvements making a difference in your quality of life.
Things you can do at home to help prevent dry eyes:
Blink more often
Avoid staring at computers and phones for an extended period. Make a conscious effort to rest your eyes in regular intervals, such as every twenty minutes. These are especially important when your daily activities require you to use smartphones and devices. Blinking can really help.
Remove eye makeup
Remove makeup like eyeliner, mascara foundation and eye shadow thoroughly.
Don’t over wear your contact lenses
Do not wear your contact lenses too long. Always keep them clean and never forget to take them out at night. Wear daily disposable lenses to prevent bacterial buildup and keep your eyes feeling fresh.
Reduce cigarette smoking
Cigarettes contain hundreds of toxic chemicals that inflame and damage your eyes. Industrial chemicals, smoke and fumes can also affect your eyes. Make sure your eyes are protected from your environment.
Home remedies
- Apply warm compresses to your eyes.
- Invest in a humidifier for your home or workplace to add moisture to the air.
- If you have an allergy that causes your eyes to itch, ask your doctor which antihistamine eye drops to use to prevent them from itching. Drops may reduce eye rubbing, so you don’t create more inflammation and dryness.
Dry Eye Treatments
The first line of treatment is usually artificial tears that give some temporary relief. These solutions and ointments do little to arrest or reverse any damaging conditions. Many brands are available without a prescription. For mild cases, try several to find the one you like best. The preservatives in some eye drops can irritate the eye, and preservative-free artificial tears may be required. Researchers are trying to develop better artificial tears formulated to normalize the electrolyte balance while lubricating the ocular surface. One promising new treatment now in clinical trials is the use of eye drops containing androgen. Nutritional supplements, such as flaxseed or fish oil capsules, may also be beneficial.
For more severe cases of dry eye, in which the cornea is inflamed, anti-inflammatory agents are sometimes prescribed. Topical steroids (in eye drops) are safe for short-term use to combat inflammation but can cause side effects when used for a long time. RESTASIS™ and XIIDRATM prescription eye medications are a valuable treatment option for many dry eye patients. RESTASIS combats dry eye disease by helping the eyes produce healthier and more abundant tears. XIIDRA is approved to treat the signs and symptoms of the disease by reducing inflammation.
Dry Eye Procedures
If eye drops and medication don’t work, procedures may be used to treat dry eyes. Treatment will be different for each person depending on the root cause of the dry eye. Some of these procedures require a series of treatments to achieve results. Maintenance sessions may also be required.
Punctal occlusion
A punctal occlusion procedure can permanently or temporarily close the tear drainage, allowing the eyes to retain the tears much longer before they evaporate. However, punctal occlusion may also increase inflammation, so your eye care professional will weigh the benefits of punctal plugs with you.
Other procedures
Other procedures are available such as intense pulsed light treatment, Lipiflow, scleral contact lenses and amniotic membranes and lid closure procedures for extreme cases of dry eye.
Dry eyes can be uncomfortable and affect your quality of life. Your doctor will review your test results, examine your eyes’ gland structure, and then discuss your options with you. Patients have vastly different therapeutic responses, so be patient and tenacious. Keep in mind that treating dry eye is a process. Even though there is no cure, there can be a solution.